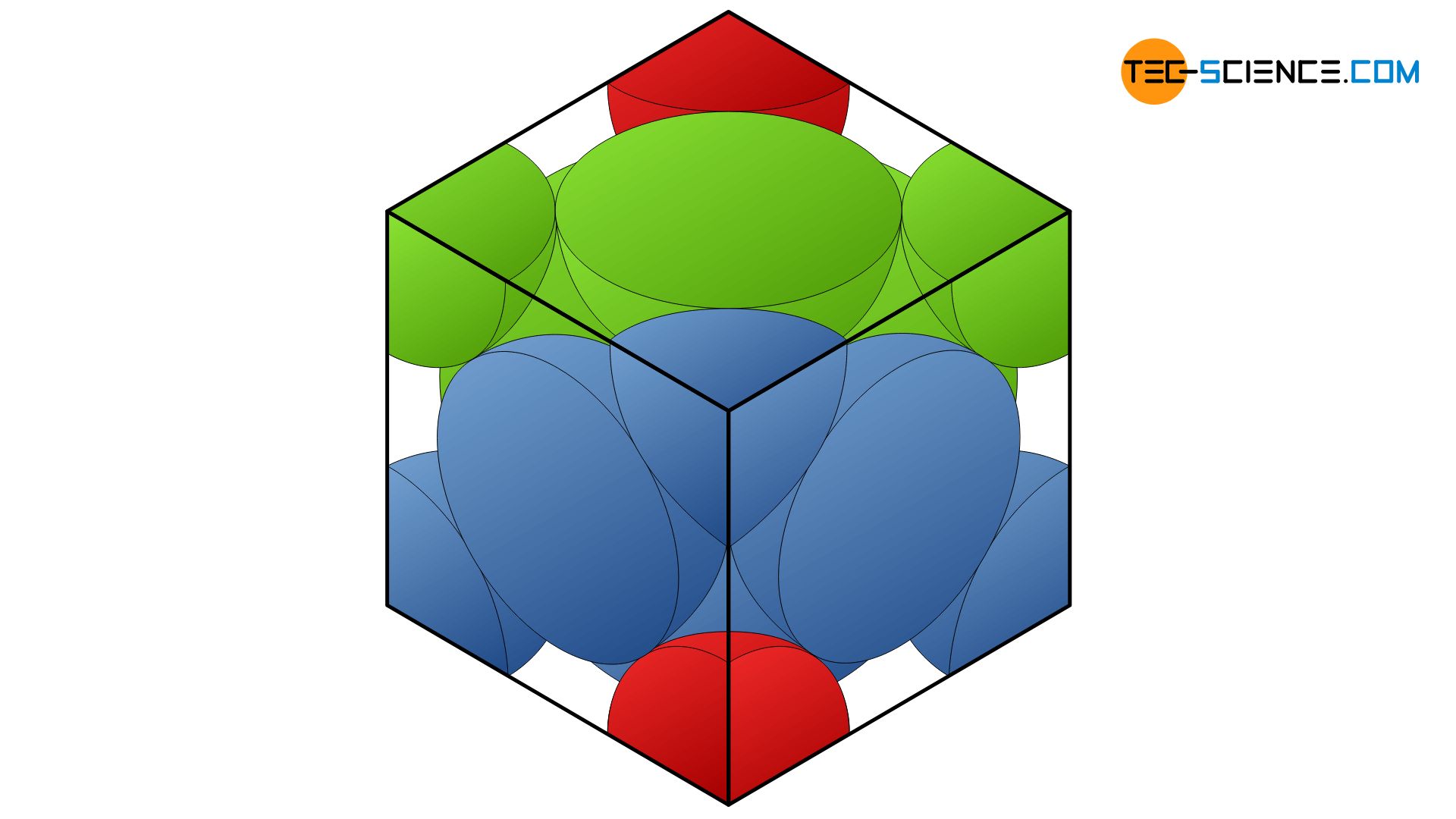
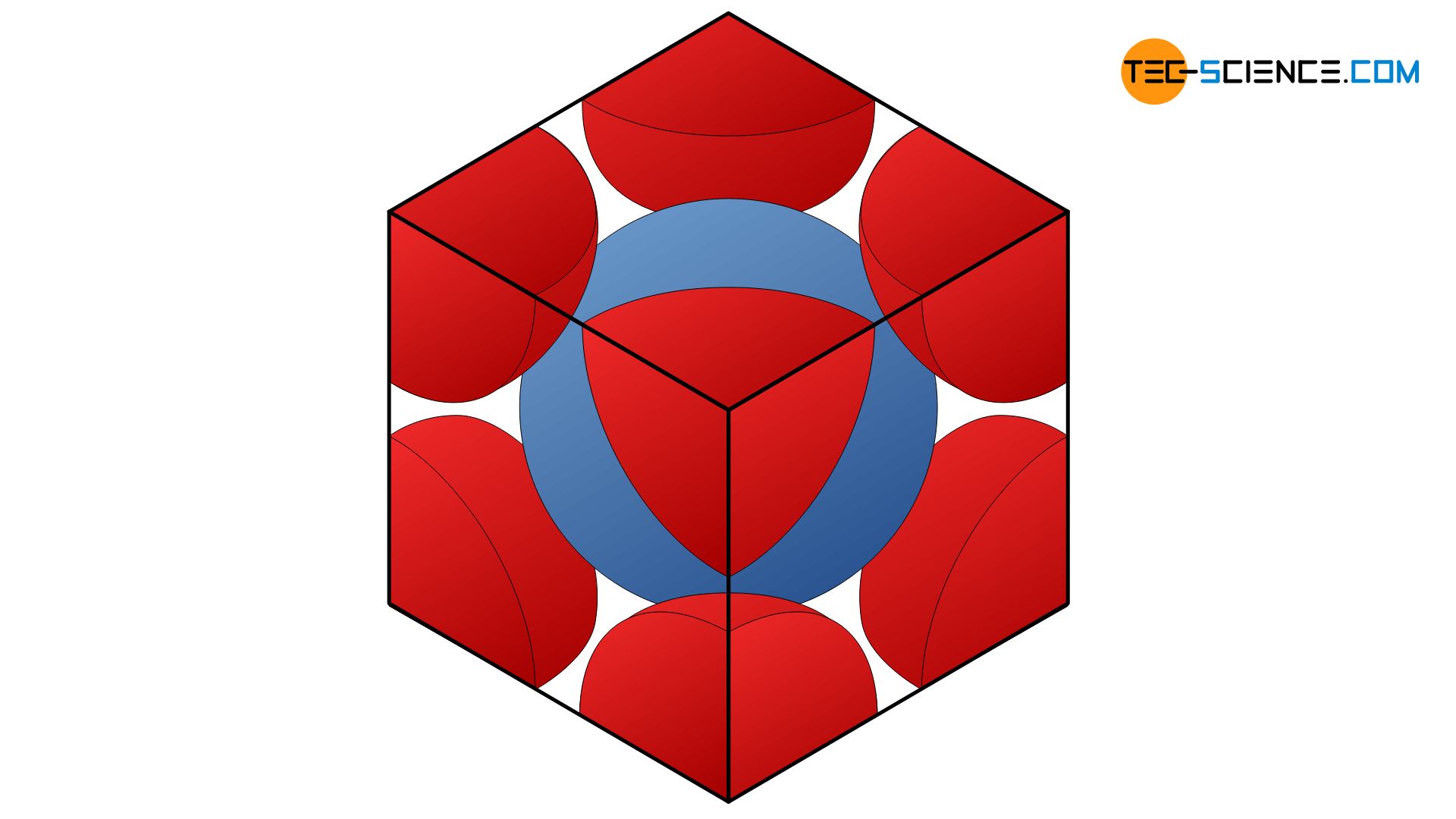
Packing efficiency of Unit Cell - Calculating the Packing efficiency of Face Centered Cubic Unit Cell, Body Centered Cubic Unit Cell and Simple Cubic Unit cell FAQs with Images.

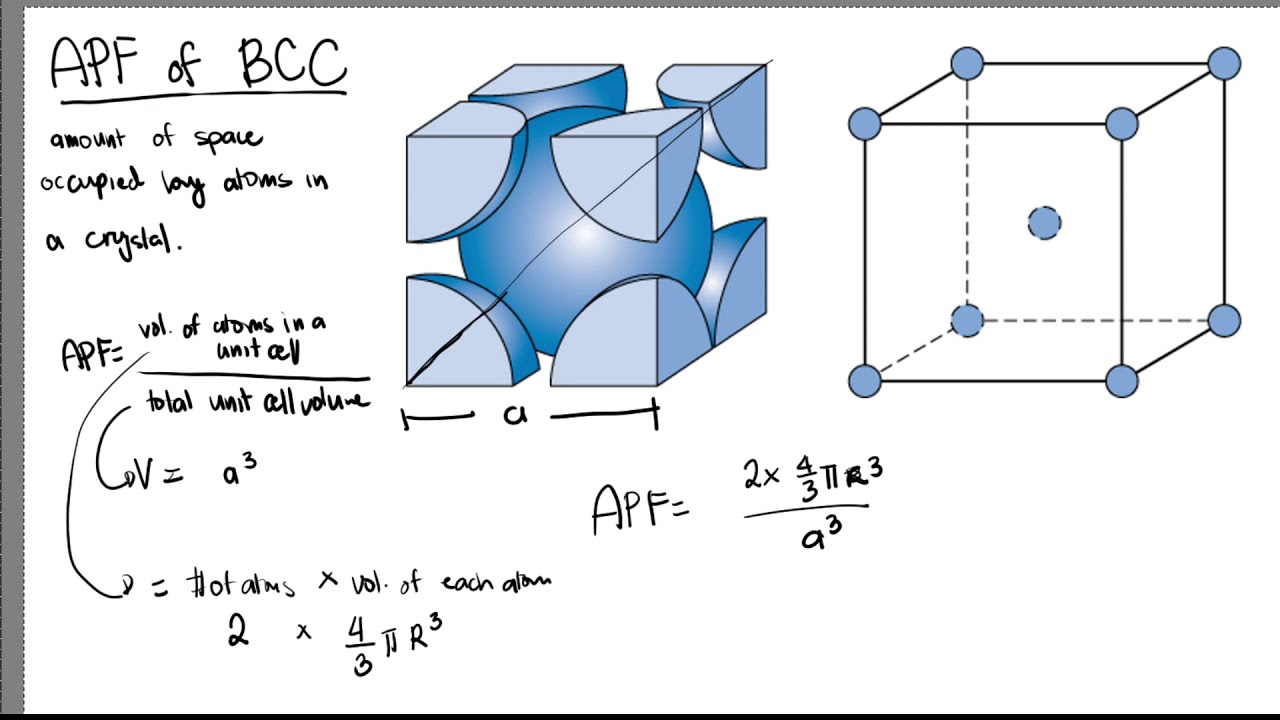
Structure of Solids Objectives By the end of this section you should be able to: Calculate atomic packing factors (HW) Compare bcc, fcc and hcp crystal. - ppt download

How can i calculate packing efficiency of bcc structure - Chemistry - The Solid State - 13423615 | Meritnation.com
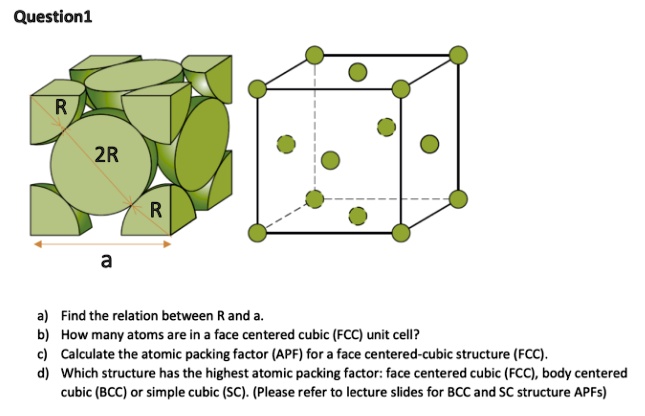
SOLVED: Question 1: Find the relation between R and how many atoms are in a face-centered cubic (FCC) unit cell. Calculate the atomic packing factor (APF) for the face-centered cubic structure (FCC).

Three-dimensional packing of spheres: bcc (Im 3m), fcc (Fm 3m) and hcp... | Download Scientific Diagram